 Therefore, anodes oxidize (lose electrons) while cathodes reduce (gain electrons). However, a cathode is referred to as the positive electrode because it gains electrons rather than loses them. Like an anode, a cathode is one of the electrodes in a battery as well. A good anode should be an efficient reducing agent, have good conductivity, stability, and a high coulombic output (the electrical energy output). These include zinc, lithium, graphite, or platinum. 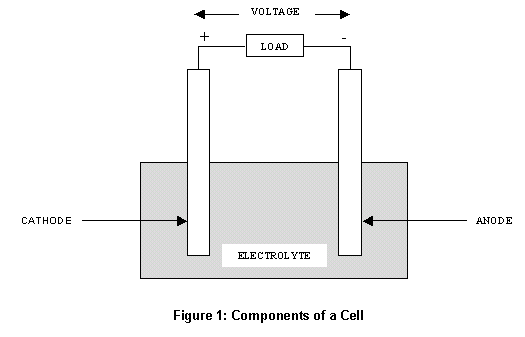
Materials Good for AnodesĪnodes can come from several different materials. 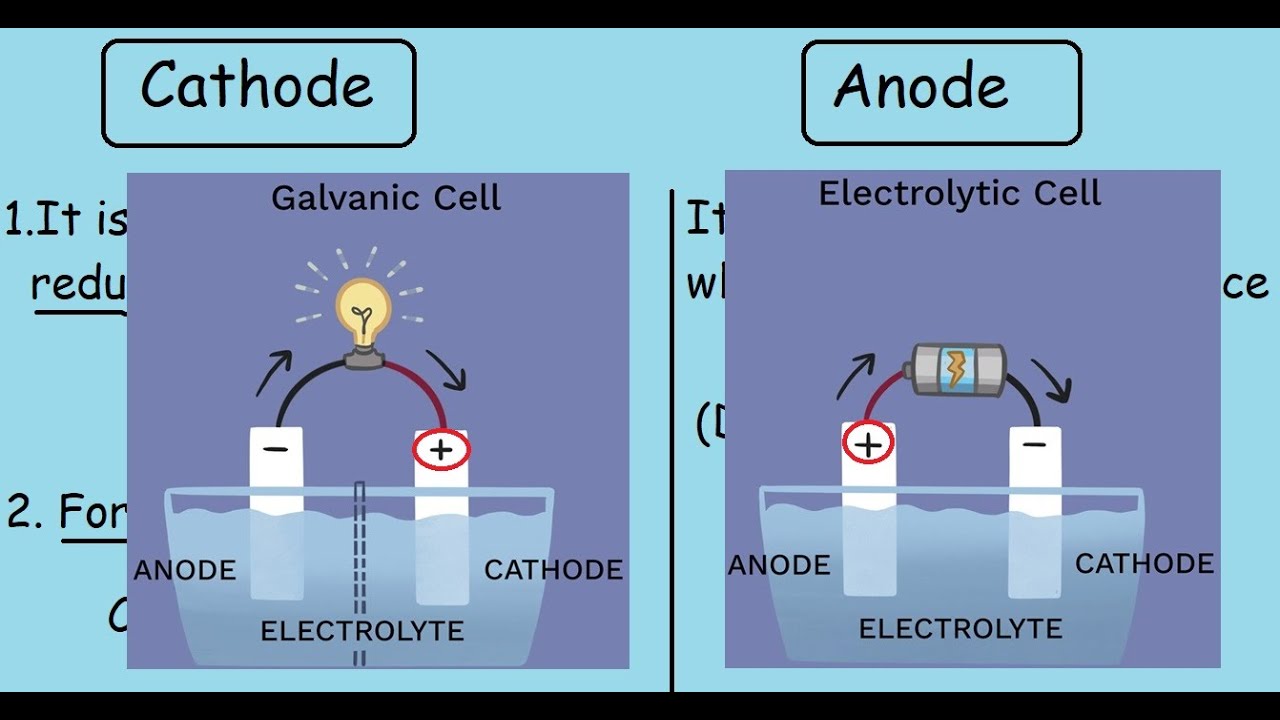
Once the anode completely erodes, the battery dies (or loses charge). The conductor (whether it be a metal wire or tube) is how we access the electricity the anode makes, and ultimately, how a battery powers our devices. It resides in an electrolyte solvent and slowly erodes as electrons move along a conductor to the cathode. How Does an Anode Work?Īn anode is an oxidizing metal, such as zinc or lithium, which means it loses electrons. This is an electrochemical reaction that produces electrons (i.e., electricity). It’s usually made of a metal that oxidizes and sends electrons to the cathode (the positive electrode). AnodeĪn anode is a negative electrode and it’s one of the essential parts of a battery. Below, we dissect the components a battery will need to charge and emit power (in a bite-sized, understandable way). Trying to understand how batteries work can feel like learning another language. Let’s get started! Anode vs Cathode in Batteries: An Overview Here we discuss what an anode is, what a cathode is, how they both work, and where they’re used. So, if you’re looking for an article that spells out the difference in plain English, look no further. Most of us rarely deal with these terms unless we’re fixing a water heater or installing your own vehicle or boat batteries. If you were today-years-old when you learned what an anode vs cathode was, you’re not alone. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
